PLEASE NOTE:
We are currently in the process of updating this chapter and we appreciate your patience whilst this is being completed.
Epidemic theory (effective and basic reproduction numbers, epidemic thresholds) and techniques for analysis of infectious disease data (construction and use of epidemic curves, generation numbers, exception reporting and identification of significant clusters)
Communicable disease control is considered in detail in a separate section of the DFPH syllabus – See Section 2G: Communicable Disease. This page covers the basic principles of epidemic theory.
Basic reproduction number (R0)
The basic reproduction number (R0) is used to measure the transmission potential of a disease. It is the average number of secondary infections produced by a typical case of an infection in a population where everyone is susceptible.1 For example, if the R0 for measles in a population is 15, then we would expect each new case of measles to produce 15 new secondary cases (assuming everyone around the case was susceptible). R0 excludes new cases produced by the secondary cases.
The basic reproductive number is affected by several factors:
- The rate of contacts in the host population
- The probability of infection being transmitted during contact
- The duration of infectiousness.
In general, for an epidemic to occur in a susceptible population R0 must be >1, so the number of cases is increasing.1
In many circumstances not all contacts will be susceptible to infection. This is measured by the effective reproductive rate (R)
Effective reproductive number (R)
A population will rarely be totally susceptible to an infection in the real world. Some contacts will be immune, for example due to prior infection which has conferred life-long immunity, or as a result of previous immunisation. Therefore, not all contacts will become infected and the average number of secondary cases per infectious case will be lower than the basic reproduction number. The effective reproductive number (R) is the average number of secondary cases per infectious case in a population made up of both susceptible and non-susceptible hosts. If R>1, the number of cases will increase, such as at the start of an epidemic. Where R=1, the disease is endemic, and where R<1 there will be a decline in the number of cases.
The effective reproduction number can be estimated by the product of the basic reproductive number and the fraction of the host population that is susceptible (x). So:
R = R0x
For example, if R0 for influenza is 12 in a population where half of the population is immune, the effective reproductive number for influenza is 12 x 0.5 = 6. Under these circumstances, a single case of influenza would produce an average of 6 new secondary cases.1
To successfully eliminate a disease from a population, R needs to be less than 1.\
Herd immunity
Herd immunity occurs when a significant proportion of the population (or the herd) have been vaccinated (or are immune by some other mechanism), resulting in protection for susceptible (e.g. unvaccinated) individuals. The larger the number of people who are immune in a population, the lower the likelihood that a susceptible person will come into contact with the infection. It is more difficult for diseases to spread between individuals if large numbers are already immune as the chain of infection is broken.
The herd immunity threshold is the proportion of a population that need to be immune in order for an infectious disease to become stable in that community. If this is reached, for example through immunisation, then each case leads to a single new case (R=1) and the infection will become stable within the population.
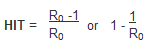
If the threshold for herd immunity is surpassed, then R<1 and the number of cases of infection decreases. This is an important measure used in infectious disease control and immunisation and eradication programmes.
Epidemics
An epidemic is defined as an increase in the frequency of occurrence of a disease in a population above its baseline, or expected level, in a given time period.2 The term is used broadly and the number of cases and time period are often unspecified. It is generally more widespread than an outbreak, which usually implies two or more epidemiologically linked cases, although the two terms have been used interchangeably. Additionally, the term has also been used to describe increasing levels of non-communicable disease, such as an ‘epidemic of cardiovascular disease.’
The definition above is general, but the term has been defined quantitatively for certain infections and a threshold is selected above which the term ‘epidemic’ is applied. For example, in England levels of influenza are routinely monitored drawing on data from GP consultations and lab diagnoses. The Royal College of General Practitioners (RCGP) has defined the baseline threshold for ‘normal seasonal activity’ in England as 30 to 200 GP consultations for influenza-like illness per week per 100,000 population. The epidemic threshold would be reached if the number of consultations surpassed 200 per week per 100,000.3
Other thresholds are used in epidemic theory. The Critical Community Size (CCS) is the total population size needed to sustain an outbreak once it has appeared, and the Outbreak Threshold is the number of infected individuals that are needed to ensure that an outbreak is unlikely to go extinct without intervention.4
Epidemic curves
An epidemic curve is a graph that illustrates the distribution of the onset of new cases of an infectious disease in relation to the onset of illness. The time interval for the onset of illness used will be determined by the incubation period (see “Definitions including: incubation, communicability and latent period; susceptibility, immunity, and herd immunity” in Section 2G for a definition of this and related terms).
Epidemic curves are a useful tool in outbreak investigations, helping to:
- Determine the type of epidemic (continuous source, point source, propagated)
- Determine the difference between the maximum and minimum incubation period
- Estimate the likely time of exposure, and thus help focus investigation on a particular time period
- Determine the incubation period in cases where the time of exposure is known
- Identify outliers (below)
It would be worth looking here for examples of different types of epidemic curve5.
Index Case and Generation Time
The original case of an outbreak is labelled as the primary case. Secondary cases contract the infection from primary cases, and tertiary cases contracted theirs from secondary cases, and so on. The index case is the term given to the first recognised case, or cases, in an outbreak. Note that the index case may not turn out to be a primary case, and the primary case of an outbreak may only be identified on further investigation, if at all. The generation time describes the duration from the onset of infectiousness in the primary case to the onset of infectiousness in a secondary case (infected by the primary case).
Exception Reporting
Infectious disease surveillance ensures that the frequency of certain diseases or symptoms are monitored. If there is an abrupt increase in the frequency of a particular disease, outside of predefined limits, it will be flagged as an “exception” and thus functions as an early indicator that further investigation is required.
Significant Clusters
A cluster, or significant cluster, is an aggregation of cases related in time or place that is suspected to be greater than the number expected (although the “expected” number may not be known). The term can relate to both communicable and non-communicable disease. Clusters can be identified using spot maps (where each case is represented on a map by a coloured dot), although such maps may show apparent “clusters” in areas that are densely populated (and thus would have a higher number of expected cases). Alternatively, maps which colour areas in different shades depending on the rate of disease in each area can be used, although if the defined areas are too large it will mask real clusters.
References
- Rothman KJ, Lash T, Greenland S. Modern Epidemiology (3rd ed.), Lippincott Williams & Wilkins, 2013
- Donaldson L, Scally G. Donaldson’s Essential Public Health (3rd ed.) CRC Press, 2009
- Goddard NL et al. Appropriateness of thresholds currently used to describe influenza activity in England. Commun Dis Public Health 2003;6(3):238–45
- Hartfield M, Alizon S. Introducing the outbreak threshold in epidemiology. PLOS Pathogens, 2013;9:e10032777
- https://med.uottawa.ca/epidemiology/ - Accessed 8/04/17
© Helen Barratt, Maria Kirwan 2009, Saran Shantikumar 2018
